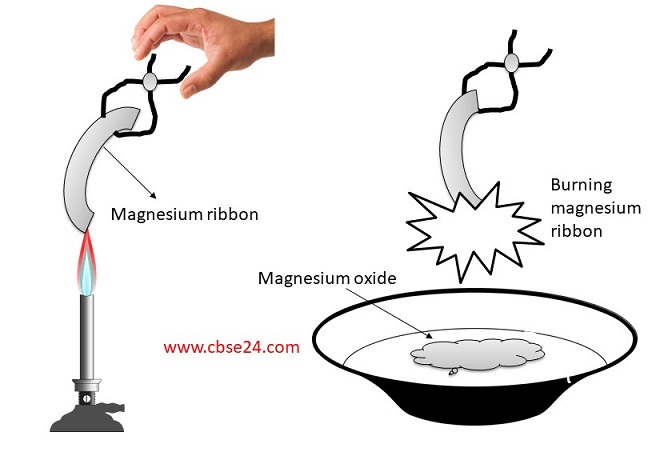
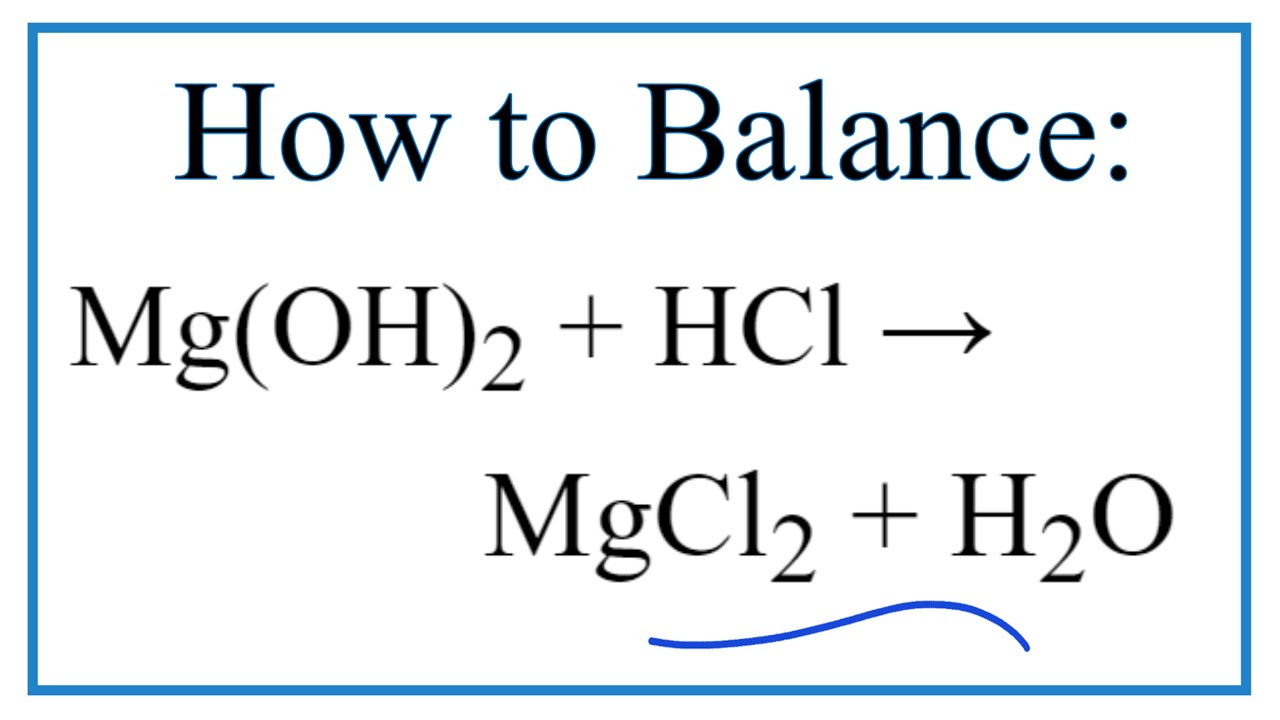
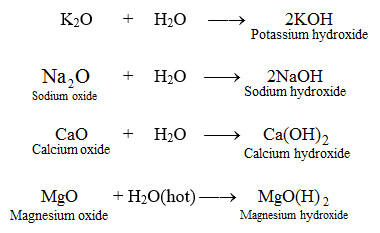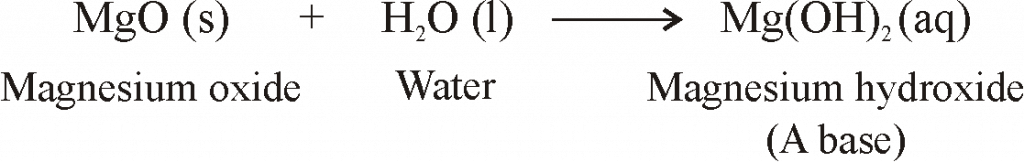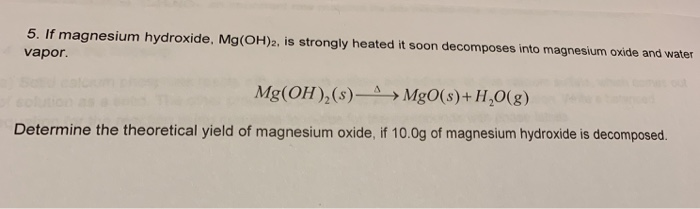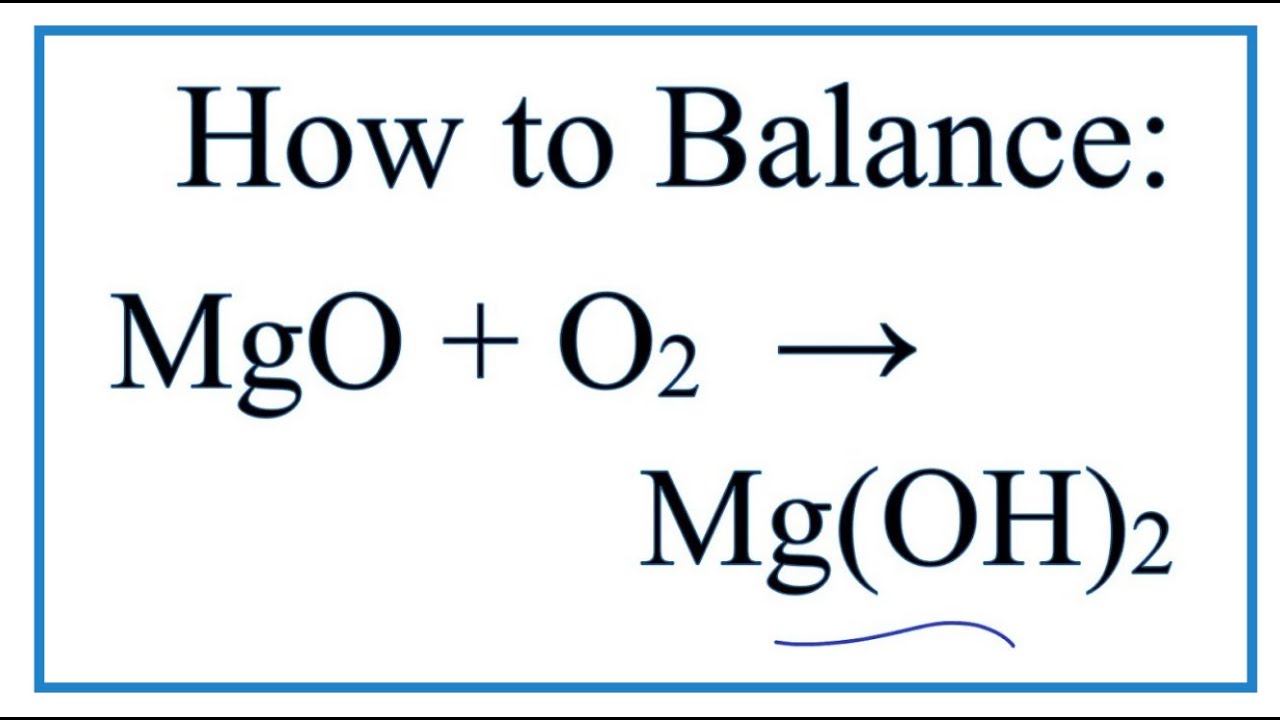MgO+ H2O=Mg(OH)2 Balanced Equation||Magnesium Oxide +Water=Magnesium hydroxide Balanced Equation - YouTube

China MgO 92% 325mesh for Water Treatment Suppliers, Producer, Manufacturers - Factory Direct Price - STAR GRACE

Water | Free Full-Text | Application of Magnesium Oxide Media for Remineralization and Removal of Divalent Metals in Drinking Water Treatment: A Review

SOLVED: In the magnesium oxide babonatory magnoium nitride ras reacted with water to form ntsium hydroxide and nnm according to the reaction given below. MgNiel + 6 H:O a Mg(OH) ( 2

Dispersive solid phase microextraction based on magnesium oxide nanoparticles for preconcentration of auramine O and methylene blue from water samples | Scientific Reports
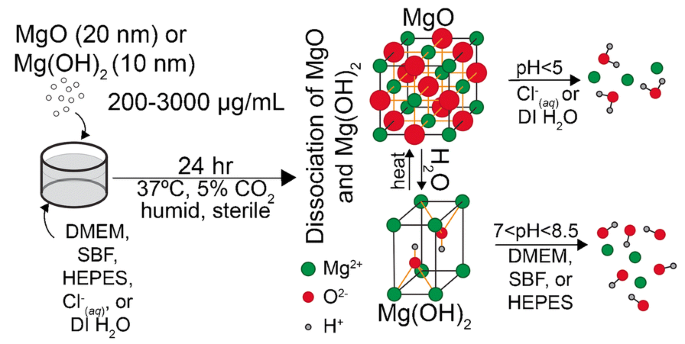
Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

What happens when Magnesium oxide is dissolved in water? Write a word equation for this process. Name - Brainly.in

Question Video: Determining the Color of Litmus Solution When Added to the Solution Made from Dissolving Magnesium Oxide in Water | Nagwa

A 12.0 gm sample of magnesium is burnt in air to form magnesium oxide (MgO) and magnesium nitrides (Mg(3)N(2)). When the products are treated with water, 1.7 gm gaseous ammonia is generated.